Patents - Philip Morris Products v Nicoventures
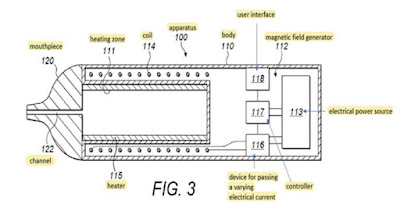
Jane Lambert Patents Court (HH Judge Hacon) Philip Morris Products SA and another v Nicoventures Trading Ltd and another [2023] EWHC 2616 (Pat) (25 Oct 2023) This was a claim by Philip Morris Products SA and Philip Morris Limited ("PMI") for the revocation of European patent (UK) 3 367 830 B1 ("EP830") and a counterclaim by Nicoventures Trading Limited and British American Tobacco (Investments) Limited ("BAT") for the infringement of that patent. There was also an application by BAT to amend the patent and an application by PMI for an Arrow declaration. These proceedings came on for trial before His Honour Judge Hacon sitting as a judge of the High Court between 21 and 23 and on 27 and 28 Sept, 2023. The learned judge delivered judgment on 25 Ocr 2023 (see Philip Morris Products SA and another v Nicoventures Trading Ltd and another [2023] EWHC 2616 (Pat) (25 Oct 2023)). By para [216] of his judgment he dismissed the counterclaim. The patent had no


.jpg)
